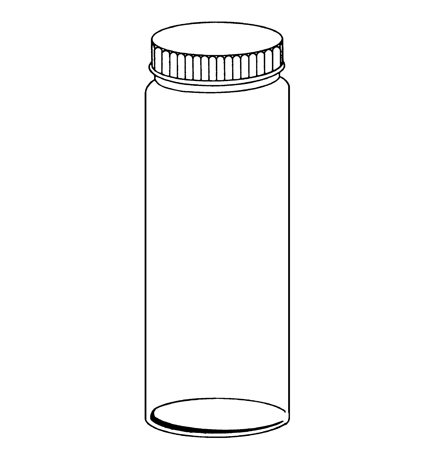
250ml Storage Bottle, #15 Threaded Top, 75mm .D (5mm wall) x 135mm OVL (5mm plated bottom) (DOB-B)
SKU: DOB-B
Price Range$21.00 – $147.20
Description
ASTM INTERNATIONAL
Designation: D1275-15
315/98
Standard Test Method for Corrosive Sulfur in Electrical Insulating Liquids1
This standard is issued under the fixed designation D1275; the number immediately following the designation indicates the year of original adoption or, in the case of revision, the year of last revision. A number in parentheses indicates the year of last reapproval. A superscript epsilon (ε) indicates an editorial change since the last revision or reapproval.
This standard has been adopted for use by government agencies to replace Method 5328-2 of Federal Test Method No. 79lb. This standard was adopted as an ASTM-IP Standard.
This standard has been approved for use by agencies of the U.S. Department of Defense.
INTRODUCTION
Prior to 2006, this test method existed as a singular method in which the main aging parameters of the test specimen were 19 h at 140°C. In 2006, it was determined that those parameters were inadequate (not sensitive enough to detect all levels of corrosive sulfur) and Method B was instituted in which the main aging parameters were 48 h at 150°C. The old parameters were kept as Method A to avoid any confusion. The current edition of this test method for the copper corrosion procedure reverts back to a singular method in which the main aging parameters are the same as the previous Method B (2006-2014). Results from the current test method cannot be compared or correlated to the method prior to 2006 and only to Method B from 2006 to 2014.
1. Scope
1.1 This test method describes the detection of corrosive sulfur compounds (both inorganic and organic) in electrical insulating liquids.
1.2 New and in-service insulating liquids may contain elemental sulfur or sulfur compounds, or both, that cause corrosion under certain conditions of use. This test method is designed to detect the presence of, or the propensity to form, free (elemental) sulfur and corrosive sulfur compounds by subjecting copper or silver to contact with an insulating liquid under prescribed conditions.
1.3 The values stated in SI units are to be regarded as the standard. Inch-pound units are included for informational purposes.
1.4 This standard does not purport to address the safety concerns, if any, associated with its use. It is the responsibility of the user of this standard to establish appropriate safety and health practices and determine the applicability of regulatory limitations prior to use.
2. Referenced Documents
2.1 ASTM Standards:2
D130 Test Method for Corrosiveness to Copper from Petroleum Products by Copper Strip Test
D923 Practices for sampling Electrical Insulating Liquids
E11 Specification for Woven Wire Test Sieve Cloth and Test Sieves
2.2 Other Document:
ANSI B74.10 rading of Abrasive Microgrits3
DIN 51 353 Testing of INsulating Oils; detection of corrosive sulfur, silver strip test4
3. Significance and Use
3.1 In most of their uses, insulating liquids are continually in contact with metals that are subject to corrosion. The presence of elemental sulfur or corrosive sulfur compounds will result in deterioration of these metals and cause conductive or high resistive films to form. The extent of deterioration is dependent upon the quantity and type of corrosive agent and time and temperature factors. Detection of these undesirable impurities, even though not in terms of quantitative values, in a means for recognizing the hazard involved.
3.2 Two methods are provided, one for copper corrosion and one for silver corrosion. Copper is slightly less sensitive to sulfur corrosion than silver but the results are easier to interpret and less prone to error. The silver corrosion procedure is provided especially for those users who have applications where the insulating liquid is in contact with a silver surface.
4. Apparatus
4.1 Bath – A hot-air oven or liquid bath provided with suitable means of heating to, and controlling at 140 or 150 ± 2°C. A circulating hot-air oven is preferred.
4.2 Bottles5 – Heavy-walled, 250-mL, bottles of chemically resistant glass constructed with necks to receive a polytetra-fluoroethylene (PTFE) threaded plug equipped with a fluoro-elastomer O-Ring. Bottles of such capacity and design are required in order to allow sufficient space for expansion of the insulating liquid and to eliminate intrusion from atmospheric gases. Flat bottomed bottles are preferred.
4.3 Copper Foil, 99.9+ % pure, 0.127 to 0.254 mm (0.005 to 0.010 in.) in thickness.
4.4 Silver Foil, 99,9+ % pure, 0.5 mm thick.
4.5 Polishing Material, consisting of 240-grit silicon carbide paper or cloth (refer to Specification E11), and also 230-mesh silicon carbide grains and pharmaceutical absorbent cotton.
Note 1 – It should be noted that 240-grit silicon carbide paper and 230-mesh silicon carbide grains have particle sizes of about the same size (63 μm). In the United States, abrasive papers are classified in accordance with ANSI B74.10. Abrasive powders are classified by ASTM mesh size.
5. Reagents
5.1 Acetone, ACS reagent grade.
5.2 Hydrochloric Acid – ACS reagent grade, 36.5 to 38.0 % assay.
5.3 Nitrogen Gas – Commercial cylinders of nitrogen gas are satisfactory for this purpose.
5.4 Suitable Solvent – technical grade acetone, heptane, hexane and pentane.
5.5 Water, distilled.
6. Summary of Test Methods
6.1 Copper Corrosion – 220 mL of insulating liquid is aged in a sealed heavy-walled bottle for 48 h at 150°C in the presence of a copper strip.
6.2 Silver Corrosion – 220 mL of insulating liquid is aged in a sealed heavy-walled bottle for 48 h at 150°C in the presence of a silver strip.
7. Preparation of Apparatus
7.1 Chemically clean bottles and PTFE screw plugs with solvents to remove insulating liquid residue, then wash the bottles with a suitable solvent such as heptane or hexane, or us a phosphate-type cleaning powder or liquid. Rinse with tap water, then with distilled water, and dry in an oven. Replace the fluoro-elastomer O-Ring before each test to avoid cross contamination.
7.2 Polish a larger piece of copper or silver foil from which, after the final polishing, several strips of the proper size may be cut. Remove blemishes from both surfaces of the copper or silver foils with the 240-grit silicon carbide paper. Cut a strip(s) of copper or silver foil 6 by 25 mm (1/4 by 1 in.). Strips may be stored in sulfur-free acetone at this point for future use. Do the final polishing of the strip by removing it from the acetone, holding it in the fingers protected with ashless filter paper or nitrile gloves, and rubbing with 230-mesh silicon carbide grains picked up from a glass plate with a pad of absorbent cotton moistened with a drop of acetone. Wipe the strip with fresh pads of cotton and subsequently handle only with stainless steel forceps (do not touch with fingers). Rub in the direction of the long axis of the strip. Clean all metal dust and abrasive from the strip, using successive clean cotton pads until a fresh pad remains unsoiled. Bend the clean strip in a V-shape at approximately at 60° angle and wash successively in acetone, distilled water, and acetone. Dry in an oven for 3 to 5 min at 80 to 100°C and immediately immerse the copper or silver strip in the prepared test specimen of insulating liquid (Note 2). Do not use compressed air or an inert gas to dry the metal strip.
Note 2 – This method of cleaning has been adapted from Test Method D130.
8. Copper Corrosion Procedure
8.1 Retrieve a sample for testing in accordance with Practices D923. Use the insulating liquid to be tested as received. Do no filter the insulating liquid.
8.2 Promptly place the prepared copper strip in a clean 250-mL bottle to which has been added 220 mL of the insulating liquid to be tested. Place the bent copper strip standing on its long edge so that no flat surface lies along the glass bottom of the vessel. Bubble nitrogen through the insulating liquid in the bottle by means of a 1.5 mm inner diameter (approximately 1/16 in. inner diameter) glass or stainless steel tube connected to the reduction or needle valve of the cylinder (connections must be sulfur-free) for 5 min at a rate of 0.5 /min. Quickly screw in the PTFE plug equipped with flouro-elastomer O-Ring (Note 3).
8.3 Place the bottle with the test specimen in the oven at 150°C, partially unscrew the PTFE plug to release the pressure and then screw it back down. REmove the bottle after heating for 48 h ± 20 min at 150 ± 2°C. Allow to cool. Carefully take the copper strip from the bottle and wash with acetone or other suitable solvent to remove all of the insulating liquid and let air dry. Do not use pressurized air to dry the copper strip.
8.4 To inspect, hold the test strip in such a manner so that light reflected from it at an angle of approximately 45° will be observed. Interpret and report the results as defined in Sections 9 and 10. If there is difficulty in determining whether the copper strip is considered to be noncorrosive or corrosive, refer to 8.5 and 8.5.1.
8.5 If a specimen is borderline or unclear (between noncorrosive and corrosive), cut the strip in half along the bent portion of the copper strip. Retain one half and treat the other half as described in 8.5.1.
8.5.1 Submerge the half of the copper strip completely in a 1:1 solution of HCl and distilled water at room temperature and wait 20 ± 2 min while gently swirling on occasion. If the deposit in question is still present and did not fade or discolor, then interpret and report the result as corrosive in Sections 9 and 10. If the deposit has been removed or has turned tan/beige, then corrosive sulfur is not present and the result is to be reported as noncorrosive. The tarnish level is then determined on the untreated portion of the copper strip. Report the tarnish level of the untreated portion of the copper strip as listed in 9.1 disregarding the color on the strip that made it unclear in the initial inspection of the strip.
Note 3 – The corrosive effects of the unstable sulfur compounds present in the insulating liquid are reduced when aged under strong oxidizing conditions such as when oxygen is present. It is imperative to keep the bottle well sealed after nitrogen bubbling. If the insulating liquid visibly darkens during testing then most likely oxygen has entered the test bottle and oxidized the insulating liquid. In the cases, the test procedure has become compromised and must be repeated.
9. Interpretation of Results
9.1 Classify the insulating liquid as corrosive or noncorrosive in accordance with Table 1. Classification of corrosive or noncorrosive is to be aided through the use of ASTM Copper Strip Corrosion Standards as referenced in Test Method D130. Often there are several different tarnish levels on a copper strip. In these cases, report the most tarnished classification. A description of the tarnish levels is provided in Table 2 for reference only.
10. Report
10.1 Report the following information:
10.1.1 Sample identification,
10.1.2 Test specimen as being corrosive or noncorrosive,
10.1.3 Tarnish level according to Test Method D130,
10.1.4 HCl treated if the strip was treated per 8.5.1, and
10.1.5 This Method, Copper Corrosion.
| TABLE 1 Copper Strip Classifications | |
|---|---|
| Classification | Description |
| Noncorrosive | Orange, red, lavender, multicolored with lavender blue or silver, or both,overlaid on claret red, silvery, brassy or gold, magenta overcast on brassy strip, multicolored with red and green showing (peacock) but no gray |
| Corrosive | Transparent black, dark gray or dark brown, graphite orlusterless black, glossy or jet black, any degree or flaking |
| TABLE 2 Copper Strip Tarnish Level Classifications, Test Method D130 | ||
|---|---|---|
| Classification | Designation | Description |
| Freshly Polished Strip | … | Unable to reproduce upon aging so no description is provided |
| 1 | Slight tarnish | a. Light orange, almost the same as freshly polished strip |
| … | b. Dark orange | |
| 2 | Moderate tarnish | a. Claret red |
| … | b. Lavender | |
| … | c. Multicolored with lavender blue or silver, or both, overlaid on claret red | |
| … | d. Silvery | |
| … | e. Brassy or gold | |
| 3 | Dark tarnish | a. Magenta overcast on brassy strip |
| … | b. Multicolored with red and green showing (peacock), but no gray | |
| 4 | Corrosion | a. Transparent black, dark gray or brown with peacock green barely showing |
| … | b. Graphite or lusterless black | |
| … | c. Glossy or jet black | |
11. Silver Corrosion Procedure
11.1 Using a silver strip, instead of a copper strip, repeat the instructions provided in 8.1 – 8.3.
11.2 To inspect after the 48 h heating, hold the test strip in such a manner so that light reflected from it at an angle of approximately 45° will be observed. Interpret and report the results as defined in Table 3 (note 4). If there is difficulty in determining whether the silver strip is considered to be noncorrosive or corrosive, refer to 11.3.
11.3 On occasion, it can be difficult to accurately determine the coloration of the silver strip and thus whether or not the insulating liquid specimen is corrosive or noncorrosive. In those cases, use energy dispersive X-ray spectroscopy (EDX or EDS) to determine the concentrations of silver and sulfur on the silver strip.
11.3.1 Mount the silver strip on an aluminum electron miroscopy stub with conductive adhesive. Do not coat the silver strip as it is already conductive. Place the silver strip into the sample chamber and apply vacuum in accordance with the manufacturer’s instructions.
11.3.2 The following parameters have been found satisfactory when performing the analysis:
11.3.2.1 Working distance: 19 to 30 mm.
11.3.2.2 Variable pressure mode at around 30 Pa or column vacuum at 1.7 x 10-7.
11.3.2.3 Accelerating voltage: variable around 20.0 kV.
11.3.2.4 Probe current: dependent on manufacturer but around 93 μA.
11.3.2.5 QC on daily basis when instrument is in use, using aluminum, copper or cobalt standards.
| TABLE 3 Silver Strip Classifications | |
|---|---|
| Classification | Description |
| Noncorrosive | Silver, yellow, golden yellow, shades of blue |
| Corrosive | Grays to black, brownish |
11.3.2.6 Perform the analysis on any discolored areas of the silver strip.
11.3.3 If the sulfur concentration of any of the discolored areas tested is 1 weight percent or greater, the result is to be reported as corrosive.
Note 4 – Table 3 is excerpted from the German Method DIN 51 353.
12. Interpretation of Results
12.1 Classify the insulating liquid as corrosive or noncorrosive in accordance with Table 3.
13. Report
13.1 Report the following information:
13.1.1 Sample identification,
13.1.2 Test specimen as being corrosive or noncorrosive,
13.1.3 Weight percentages of silver and sulfur if applicable, and
13.1.4 This Method, Silver corrosion.
14. Precision and Bias
14.1 No statement is made about either the precision or bias of this test method since the result merely states whether there is conformance to the criteria for success specified in the procedure and is not quantitative.
14.2 Results from repeatability testing from one laboratory are provided in Table 4 and Table 5.
15. Keywords
15.1 copper; corrosion; corrosive sulfur; elemental sulfur; free sulfur; insulating; insulating liquid; mineral; natural ester; oils; petroleum; silicone; silver; sulfur; synthetic ester
| TABLE 4 Repeatability on the Copper Corrosion Procedure, Noncorrosive or Corrosive | |||
|---|---|---|---|
| Sample | Concentration in Test Specimen <1 mg/kg Elemental Sulfur | Concentration in Test Specimen <8 mg/kg Elemental Sulfur | Concentration in Test Specimen <80 mg/kg Elemental Sulfur |
| 1 | noncorrosive | corrosive | corrosive |
| 2 | noncorrosive | corrosive | corrosive |
| 3 | noncorrosive | corrosive | corrosive |
| 4 | noncorrosive | corrosive | corrosive |
| 5 | noncorrosive | corrosive | corrosive |
| 6 | noncorrosive | corrosive | corrosive |
| 7 | noncorrosive | corrosive | corrosive |
| 8 | noncorrosive | corrosive | corrosive |
| 9 | noncorrosive | corrosive | corrosive |
| 10 | noncorrosive | corrosive | corrosive |
1 This test method is under the jurisdiction of ASTM Committee D27 on Electrical Insulating Liquids and Gases and is the direct responsibility of Subcommittee D27.06 on Chemical Test.
Current edition approved Nov. 15, 2015. Published January 2016. Originally approved in 1953. Last previous edition approved in 2006 as D1275 – 06 which was withdrawn January 2015 and reinstated in December 2015. DOI: 10.1520/D1275-15.
2For referenced ASTM standards, visit the ASTM website, www.astm.org, or contact ASTM Customer Service at service@astm.org. For Annual Book of ASTM Standards volume information, refer to the standard’s Document Summary page on the ASTM website.
3Available from American National Standards Institute, 25 W. 43rd St., 4th Floor, Bew York, NY 10036, USA.
4Available from Deutsches Institut für Normung e.V.(DIN), Am DIN-Platz, Burggrafenstrasse 6, 10787 Berlin, Germany, https://www.din.de or https://www.bleuth.de
5The sole source of supply of the Bottles and PTFE screw plugs known to the committee at this time is Prism Research Glass, P.O. Box 14187, Research Triangle Park, NC 27709, part number DOB-B-250.
| Name | SKU | Price | Buy |
|---|---|---|---|
| 250ml Storage Bottle, #15 Threaded Top, 75mm .D (5mm wall) x 135mm OVL (5mm plated bottom) (DOB-B) - Options: #15 PTFE Plug, Front Seal, Viton O-Ring - 10 Pack | DOB-217-112 | $21.00 | |
| 250ml Storage Bottle, #15 Threaded Top, 75mm .D (5mm wall) x 135mm OVL (5mm plated bottom) (DOB-B) - Options: Complete with TFE Front Seal Plug | DOB-B-250 | $147.20 | |
| 250ml Storage Bottle, #15 Threaded Top, 75mm .D (5mm wall) x 135mm OVL (5mm plated bottom) (DOB-B) - Options: Bottle Only | DOB-B-250a | $107.47 |